A Smarter Approach to CAUTI Prevention with Spigot Guard
Innovative Solutions for Safer Healthcare
Foley catheter contamination prevention
Best Practice. Made Better.™
An important clinical component of reducing CAUTIs consists of the proper care and maintenance of the foley catheter system.
Current clinical practice calls for an internal and external decontamination of the drainage spigot with an alcohol prep pad after emptying the bag. This ineffective, honor-system approach is rarely performed, increasing the risk of CAUTIs. Spigot Guard is a device that improves patient outcomes by improving the handling of the drain tube, promoting high-quality infection control and prevention.
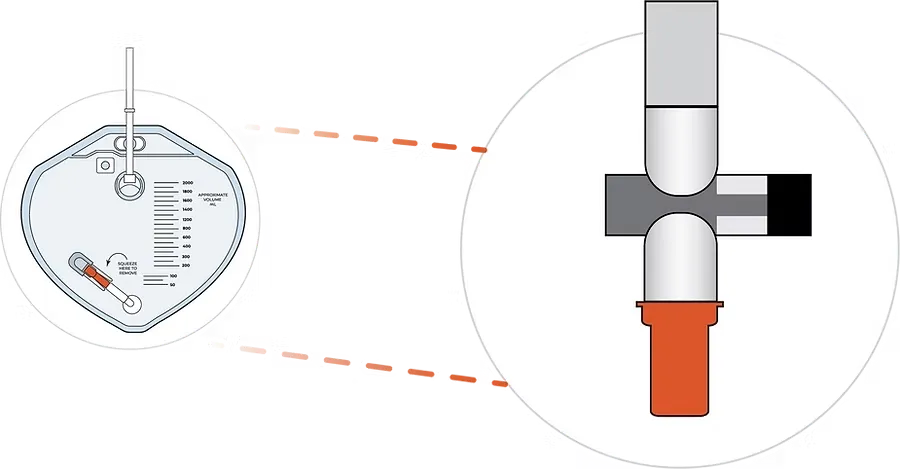
Disinfects in 1 minute. Maintains protection as long as the cap remains in place.
Healthcare givers are frontline performers in the effort to reduce health-care related infections. Yet a post-Covid hospital culture has led to suboptimal adherence to infection control practices, including the protection and maintenance of the foley catheter system. Spigot Guard provides caregivers with a highly visual, contextual cue and convenient packaging design to promote easy adherence to best practice, resulting in reduced CAUTI rates.
Protecting Patients, Empowering Nurses, Reducing Infections
Healthcare givers are frontline performers in the effort to reduce health-care related infections. Yet a post-Covid hospital culture has led to suboptimal adherence to infection control practices, including the protection and maintenance of the foley catheter system. Spigot Guard provides caregivers with a highly visual, contextual cue and convenient packaging design to promote easy adherence to best practice, resulting in reduced CAUTI rates.
Foley catheter closed system protection
Disinfects external and internal surfaces
Provides rapid disinfection of the drain surfaces, with a kill time after one minute of placement.

Creates a complete bacterial barrier
Serves as a single-use solution, ensuring full protection between drain access.

Universal fit and versatile application
Compatible with most urine collection bags, suitable for use in acute care, long-term care, and home settings.
Provides visual compliance
Spigot Guard’s bright orange color helps caregivers verify that a port is clean at a glance.
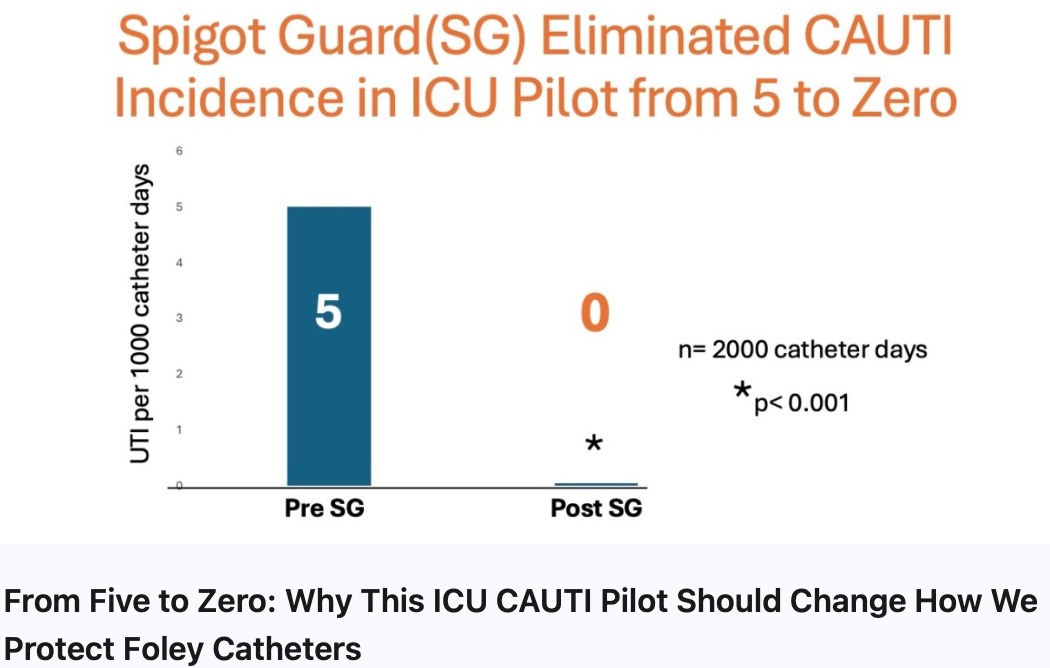
CAUTI prevention pilot program
Spigot Guard is 99.99% effective in eliminating the six most common organisms associated with CAUTIs.*
The effectiveness of Spigot Guard antiseptic caps was tested in vitro against:*
Proteus
Klebsiella
Escherichia coli
Staphylococcus aureus
Staphylococcus saprophyticus
Pseudomonas aeruginosa
Study conclusion:
All test samples exceeded the minimum 4-log reduction after one minute.
ROI CALCULATOR
Calculate your real-world CAUTI ROI with Spigot Guard
- Hospital Parameters
- Total Licensed Beds
- 500
- Occupancy Rate
- 85%
-
Catheter Rate
(% of occupied beds)
- 20%
-
CAUTI Rate
(per 1,000 catheter-days)
- 5
- Cost per CAUTI
- $12,000
- Spigot Guard Cost per Unit
- $2
- Caps per Patient per Day
- 3
- Current Hospital Profile
-
Occupied Beds
(855 occupancy)
- 425
-
Daily Catheter Patients
(20% of occupants have a foley)
- 85
- Annual Catheter-Days
- 31,025
- Cap Usage
- Daily Caps Used
- 255
- Annual Caps Used
- 93,075
- CAUTI Analysis
- Current Total CAUTIs (Annual)
- 155
- Intraluminal CAUTIs Prevented (34% × 100%)
- 53
- Annual Financial Impact
- Gross Savings(CAUTIs Prevented × Cost)
$632,910
- Gross Savings(CAUTIs Prevented × Cost)
$186,150
- Gross Savings(CAUTIs Prevented × Cost)
$446,760
Return on Investment (ROI)
Does the drain really get dirty?
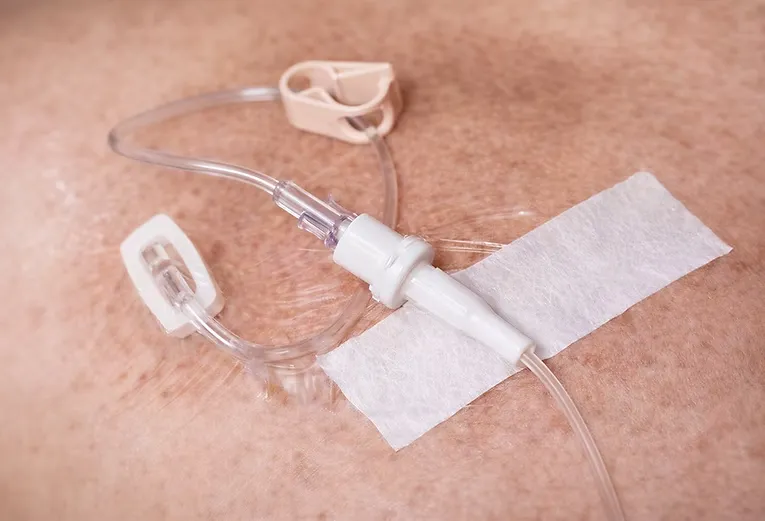
Infection prevention for urinary drainage systems
Understanding the Intraluminal Route: What the Research Shows
Skeptics sometimes question whether intraluminal contamination—bacteria entering through the catheter drainage system and ascending to the bladder—represents a significant infection source. The peer-reviewed research confirms this pathway is real, and its prevalence varies based on catheter management practices.
Two Key Studies Tell the Story
The landmark Mayo Clinic study (Tambyah et al., 1999) followed 1,497 newly catheterized patients with daily paired cultures designed to distinguish infection pathways. Among cases where the route could be determined, 34% were intraluminal—originating from drainage system contamination.
A 2021 French ICU study (Aumeran et al.) found only 11.5% intraluminal infections. The difference? This ICU achieved a 3% disconnection rate versus 10% in the Mayo study. Strict closed-system adherence reduced intraluminal infections—but in typical hospital settings with less rigorous maintenance, rates approach or exceed the Mayo finding.
Why Duration Increases the Risk
The intraluminal threat compounds over time. Every catheter day brings additional manipulations: bag changes, patient transport, specimen collection, bag emptying. Each event creates contamination opportunities. Research confirms that CA-bacteriuria correlates with catheterization duration, and the intraluminal pathway becomes increasingly significant with extended use. Once bacteria enter the lumen, they form biofilm and migrate upward toward the bladder through retrograde ascension—a mechanism confirmed through molecular tracing stud
The Bottom Line
Even with good closed-system practices, intraluminal contamination accounts for a meaningful percentage of CAUTIs. In real-world conditions, this pathway will cause one-third or more of infections. Protecting the catheter lumen addresses a documented infection route that existing protocols cannot fully eliminate
We’ve included studies below so you can review the methodology and findings yourself. The science speaks clearly—intraluminal ascension is real, measurable, and preventable.

Request a Pilot / Evaluation
Request a facility or unit-level evaluation and we’ll provide the pilot evaluation framework and protocols

Contact SpigotGuard
Request General Information and we will contact you shortly to followup